BONE-INVOLVED CANCERS (ALMB-0168)
OSTEOSARCOMA AND SOLID TUMOR METASTASES
ALMB-0168 has received Orphan Disease Drug and Rare Pediatric Disease designations from the FDA for its use in osteosarcoma. A Phase I clinical trial has been approved and started in 2020.

Bone cancers can either originate in the bone, such as osteosarcoma, or travel to the bone from tumors originating in a other location, such as breast cancer bone metastases.
Osteosarcoma is a rare cancer that primarily strikes people between the ages of 10 and 30. Most often it occurs in the growth plates of the long bones, such as the legs and arms. The surgical removal of these tumors is the best treatment option and can result in severe disfigurement and disability as the adolescent continues to grow and develop.

Most cancers are solid tumors in soft tissues (breast, prostate, etc.) and one of the most likely sites for metastases is the bone due to its fertile microenvironment. For example, there are approximately 150,000 Stage IV breast cancer patients in the US and over 50% of them will develop bone tumors. The bone metastases are often not candidates for surgical removal and can cause significant pain for the patient.
There are three major cell types in bone tissues:
- Osteocytes
- Osteoblasts
- Osteoclasts
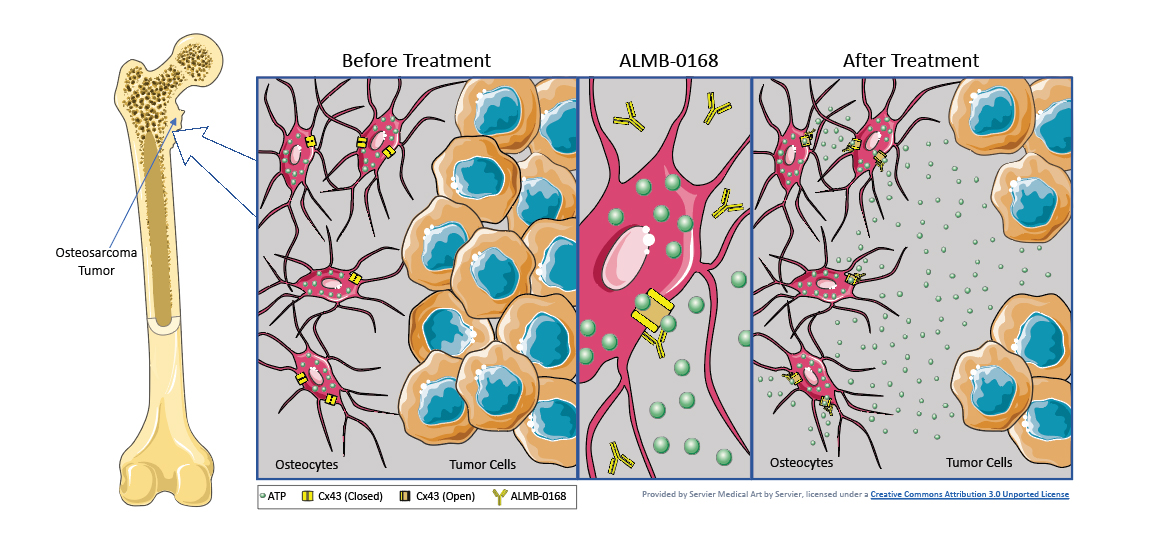
Osteocytes comprise over 95% of total bone cells and they richly express connexin43 hemichannels. These hemichannels are normally closed but, when activated to open, can release several factors, including adenosine triphosphate (ATP). ALMB-0168 activates the connexin43 hemichannels causing them to open and release ATP, as well as other molecules, into the environment outside the cells. The local increase in ATP inhibits the growth and spread of the tumor cells in the bone.
AlaMab would welcome the opportunity to discuss possible investment or partnering opportunities.
